Durham, NC – Intraventricular hemorrhage (IVH) – or bleeding in the brain – is a devastating condition common to premature babies, especially those born more than 10 weeks early. Injuries to the brain induced by severe IVH and the ensuing pressure caused by fluid buildup (known as post-hemorrhagic hydrocephalus, or PHH) can result in seizures, cerebral palsy, developmental retardation and an increased mortality rate. There is currently no effective treatment for IVH, but a study released today in STEM CELLS Translational Medicine provides information that might change that.
The study, by researchers at Sungkyunkwan University in Seoul, shows how extracellular vesicles (EVs) derived from mesenchymal stem cells (MSCs) can ease IVH-induced brain injuries. It also reveals that a protein-coding gene called brain-derived neurotrophic factor (BDNF), secreted by MSCs and transferred through EVs, is likely the key factor behind these beneficial effects.
MSCs are multipotent adult stem cells present in umbilical cord, bone marrow, fat, dental and other body tissues. Their ability to secrete biologically active molecules that exert beneficial effects on injured tissues makes them a promising target in regenerative medicine. Recent studies and clinical trials have shown that MSC-derived EVs (membranes that contain proteins, lipids and RNAs) can safely recreate the therapeutic efficacy of parent MSCs in various neonatal disorders. Moreover, as these MSC-derived EVs are a cell-free therapy, they bypass many concerns associated with MSC transplantation, such as the potential to form tumors or cause pulmonary embolisms, or a chance of the cells being rejected.
“This all has led to the questions of whether MSC-derived EVs might serve as a surrogate for stem cell therapy in cases of severe IVH and, if so, whether the neuroprotection is related to protein transfer from EVs to the damaged host brain tissue,” said the current study’s corresponding author, Won Soon Park, M.D., Ph.D.
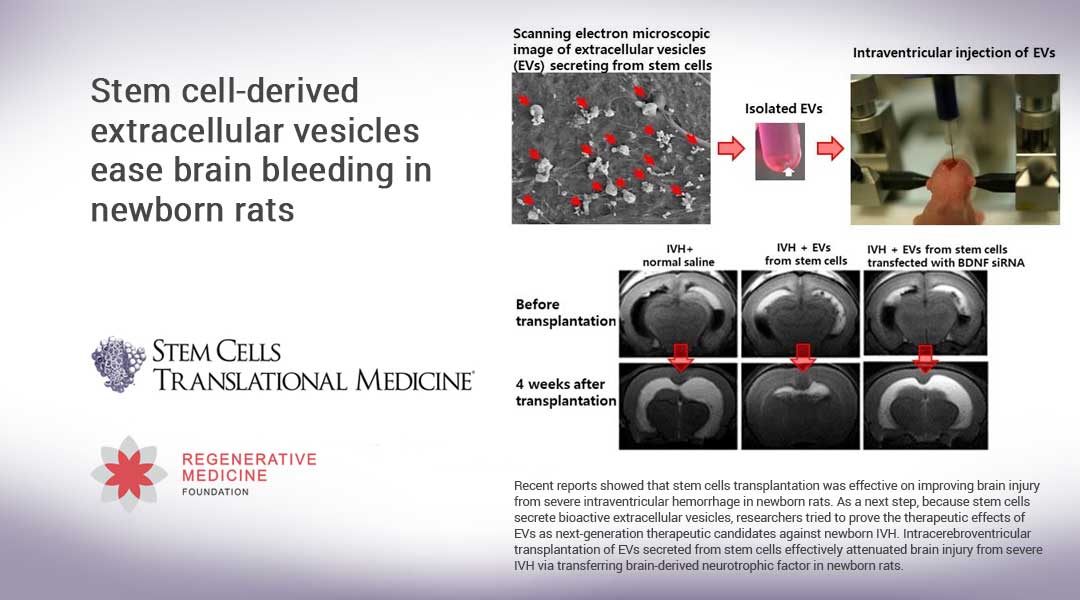
To find the answers, they applied either MSCs, MSC-derived EVs with or without BDNF knockdown, or fibroblast-derived EVs in vitro to rat neuronal cells that had first been exposed to thrombin to induce cell death. They repeated the process in vivo by injecting 4-day-old rats with 200 μL of blood to induce severe IVH, and then two days later treating them with a transplantation of either MSCs or one of the EV groups. (A non-treated group of newborn rats was used as a control.) The results were assessed via MRIs and functional behavioral tests.
“We learned that the MSCs and MSC-derived EVs – but not the EVs derived from BDNF-knockdown MSCs or fibroblasts – significantly reduced both the thrombin-induced neuronal cell death in vitro and the severe IVH-induced brain injuries and inflammatory responses in vivo,” Dr. Park said. “As such, our data indicate that MSC-derived EVs are as effective as parental MSCs in attenuating severe IVH-induced brain injuries, and this neuroprotection is primarily mediated by BDNF transfer via EVs. We hope now to move on to establishing the optimal timing, route and dosage of the MSC-derived EVs.”
“The promising potential of mesenchymal stem cell derivatives are highlighted in this early study for a treatment for bleeding in the brain, a common occurrence in premature babies,” said Anthony Atala, M.D., Editor-in-Chief of STEM CELLS Translational Medicine and director of the Wake Forest Institute for Regenerative Medicine. “This approach offers hope someday of a treatment to prevent or reduce the devastating effects of brain hemorrhage that can cause seizures, developmental delay and death in these vulnerable babies.”
###
The full article, “Brain-derived neurotropic factor mediates neuroprotection of mesenchymal stem cell derived extracellular vesicles against severe intraventricular hemorrhage in newborn rats,” can be accessed at https:/
About STEM CELLS Translational Medicine: STEM CELLS Translational Medicine (SCTM), co-published by AlphaMed Press and Wiley, is a monthly peer-reviewed publication dedicated to significantly advancing the clinical utilization of stem cell molecular and cellular biology. By bridging stem cell research and clinical trials, SCTM will help move applications of these critical investigations closer to accepted best practices. SCTM is the official journal partner of Regenerative Medicine Foundation.
About AlphaMed Press: Established in 1983, AlphaMed Press with offices in Durham, NC, San Francisco, CA, and Belfast, Northern Ireland, publishes two other internationally renowned peer-reviewed journals: STEM CELLS® (http://www.
About Wiley: Wiley, a global company, helps people and organizations develop the skills and knowledge they need to succeed. Our online scientific, technical, medical and scholarly journals, combined with our digital learning, assessment and certification solutions, help universities, learned societies, businesses, governments and individuals increase the academic and professional impact of their work. For more than 200 years, we have delivered consistent performance to our stakeholders. The company’s website can be accessed at http://www.
About Regenerative Medicine Foundation (RMF): The non-profit Regenerative Medicine Foundation fosters strategic collaborations to accelerate the development of regenerative medicine to improve health and deliver cures. RMF pursues its mission by producing its flagship World Stem Cell Summit, honouring leaders through the Stem Cell and Regenerative Medicine Action Awards, and promoting educational initiatives.